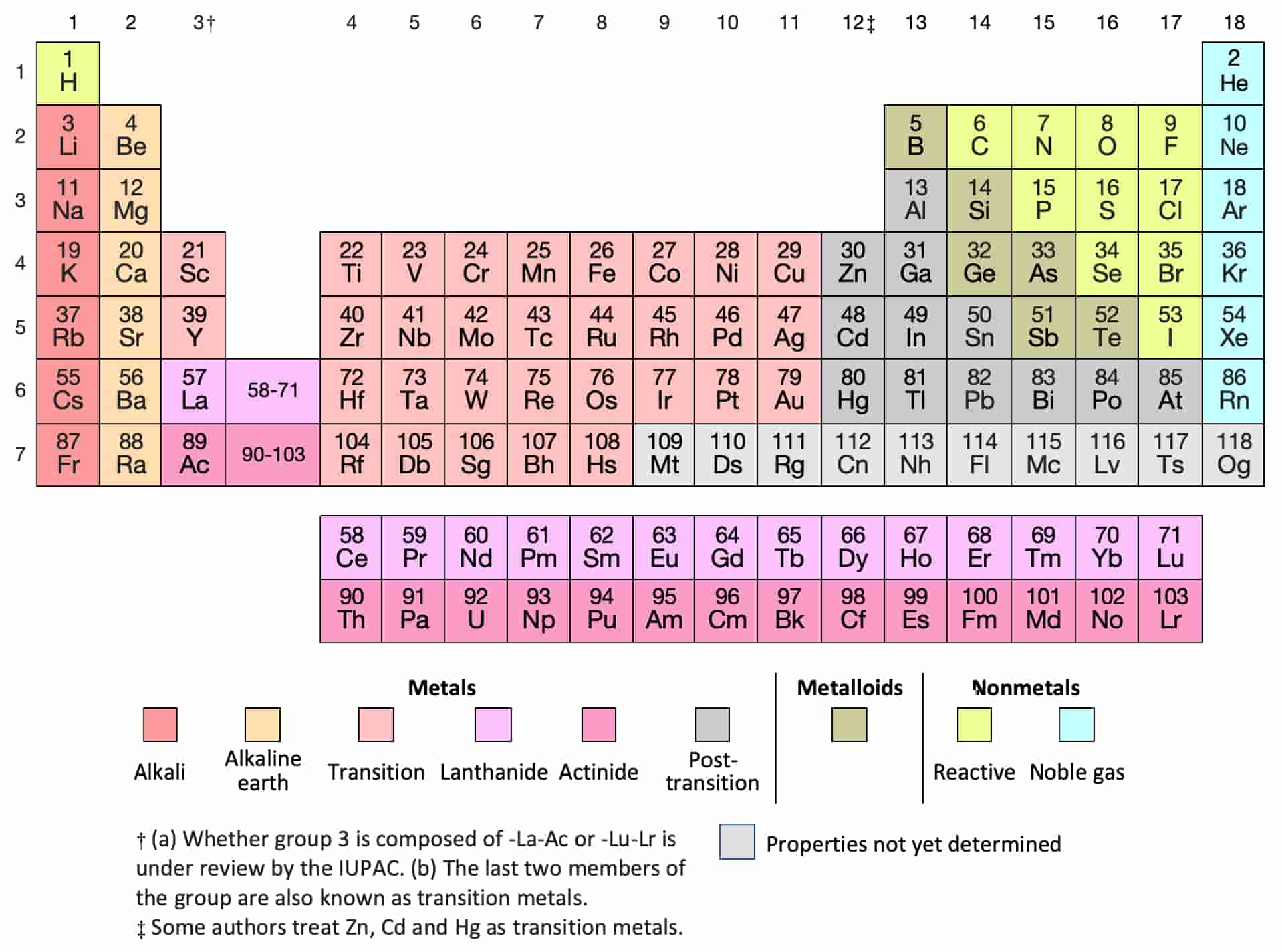
Going down the group, the color of elements increase due to decreasing effective nuclear charge, atomic radius increase, and electronegativity decreases. Halogens always exist as diatomic molecules (live in pairs). For this reason, in most chemical reactions, halogens tend to gain one electron. Many of them can be used as semiconductors.Īll halogens are missing just one electron to fill their valence electron shell. The elements around this partition have intermediate metal- and non-metal-like properties. Metalloids roughly form a staircase line in the periodic table. 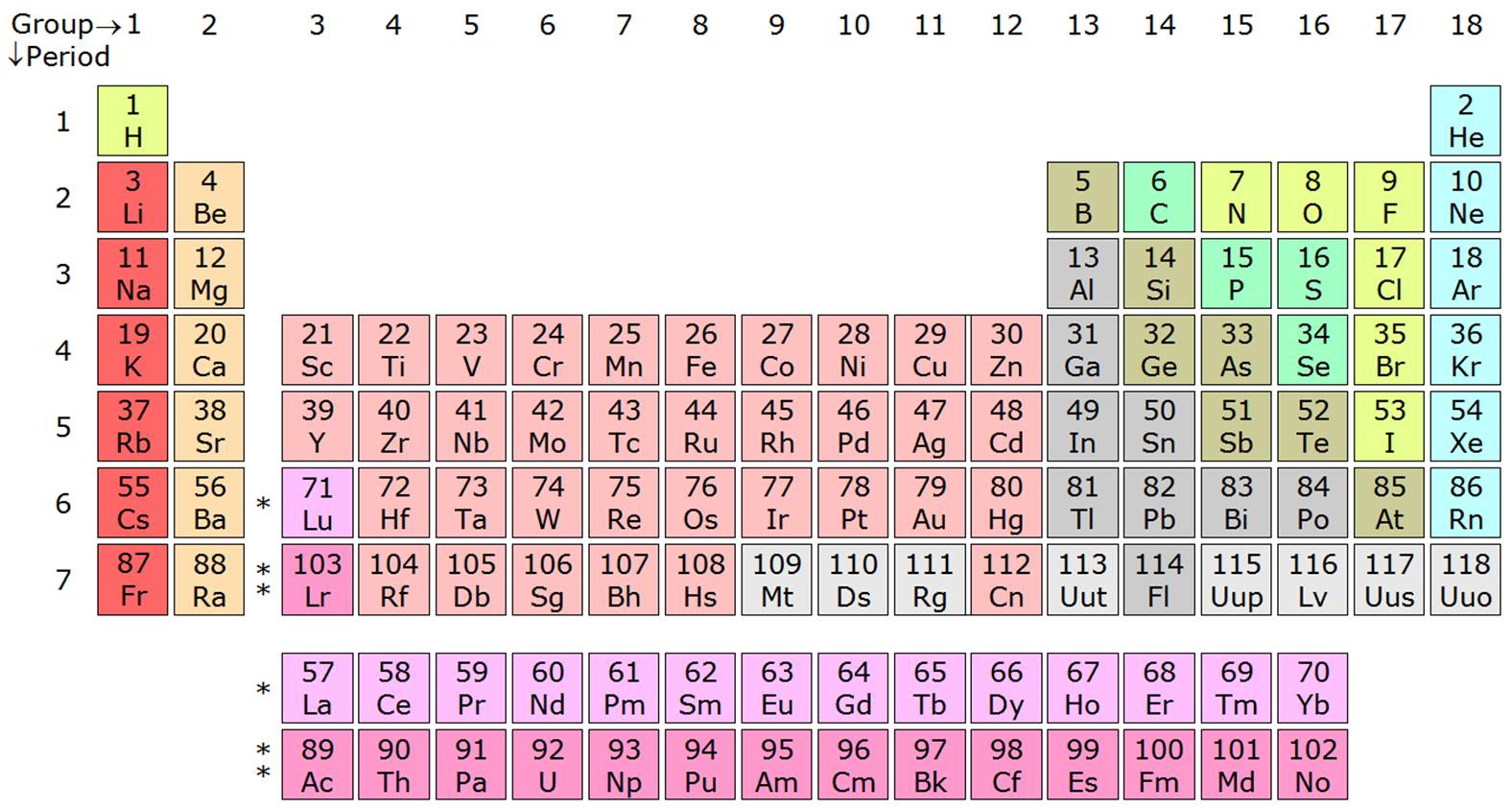
The poor metals consist of aluminum, gallium, indium, thallium, tin, lead, and bismuth. Their radioactivity is due to the fact that any nucleus with greater than 82 protons cannot be stable. The Actinidies metals - Metallic elements with atomic numbers #89-103 that fill 5f orbitals all are very radioactive, such as Uranium.The Lanthanides metals - Shiny (luster), metallic elements with atomic numbers #57-71 that fill 4f orbitals.The Inner transition metals - the separated rows at the bottom of the periodic table, "f block", are mostly synthetic: This group contains many well-known metals, such as iron (Fe), nickel (Ni), copper (cu), Chromium (Cr), mercury (Hg), and Gold (Au). The d orbitals of these elements are being filled. These metals have varying valence electrons, can have more than 1 oxidation state, and are always positive. The atoms of the transition metals have more complicated electron arrangements than other atoms. According to Moseley's Period table, These elements are found in 2nd Group. And this is also a reason for its lowest ionisation energy.Īlkaline-Earth Metals Īlkali-Earth Metals have the second lowest ionization energy. In the electron configuration of Francium the valence electron is at very far distance so that's why electron need very less energy to emit that electron (due to less attraction force). Francium reacts easily due to its low ionization energy. Reactivity increases down the group, which explains why Francium is the most reactive metal. They tend to lose 1 electron to complete their outer shells. Their reactivity is attributed to the low ionization energy of the outermost electron in the atom (one valence electron). This can be especially observed by their reaction with water, for example. These elements tend to gain electrons when forming a bond with metals or share electrons between two non-metals to form a covalent bond.Īlkaline metals are known as the most reactive metals. Non-metals are found on the top-right corner of the p-block elements. By the time you get to Neon, the electron shells are completely full! So, Lithium has one electron on the outer shell while Beryllium has two electrons on its outer shell (while the 1st shell is full). As you read across the period, the elements in the period increase with electrons on their electron shell (they do not increase in electron shell if you go left to right on a period in the periodic table, only the electrons on the electron shell increase). While the second period have elements that have two electron shells (the first electron shell contains two electrons, and the second shell can hold up to eight). For example, the first period consists of Hydrogen and Helium, which both have one electron shell. Elements in the same group act similarly because they have the same valence electron configuration.Įach elements in the same period have the same number of electron shells. Elements from the same group are found in vertical columns. The periodic table is arranged into groups (columns) and periods (rows).Įlements in the same group, or family, have similar chemical properties. Properties of elements change in systematic ways throughout the periodic table. Rows, or periods, help list elements in order of mass and atomic number. There are 18 columns that divide the elements into groups with closely related chemical properties. The periodic table arranges the elements according to their atomic number and fits them into a logical pattern (made by Henri Moseley in 1910), while when Russian scientist Dimitri Mendeleev (1870) created the periodic table, he arranged it by atomic mass. Many of them are naturally occurring, but some were made in a laboratory and are not present in nature. As of 2016, following the IUPAC's (International Union of Pure and Applied Chemists) naming of nihonium, moscovium, tennessine, and oganesson (elements 113, 115, 117, and 118 respectively), there are 118 elements currently known and officially named. The periodic table lists all the known elements. Cells with text in red are gaseous at room temperature.Ĭells with text in green are liquid at room temperature.Ĭells with text in black are solid at room temperature.Ĭells with a dashed red outline are not found naturally on earth.Įlements 43, 61, and 84 and greater are only known as radioactive.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
